Circular business models and downstream value chain collaboration in the medical device industry
Congratulations to Vita van den Broek, Master Student Innovation Management at the TU Eindhoven, who recently graduated! Her graduation project is called ‘Circular Business Models and Downstream Value Chain Collaboration in the Medical Device Industry’.


In her graduation project, Vita conducted a case study on high-value single-use devices from the perspective of the original equipment manufacturer – our valued partner, Medtronic. She developed two tools: one focused on external collaboration, and another designed as a questionnaire that allows a product to be ranked and help determine which Circular Business Model is the best fit for this product.
During her time at Medtronic, Vita was mentored by Janny Boekhout, Sustainability Manager. Janny: “Vita’s master’s thesis not only provided clear analyses, but also resulted in practical recommendations that we can genuinely build on. Vita designed two “tools.” The first focuses on (external) collaboration: a set of question cards (Stakeholder Alignment Tool). The second is an Excel-based questionnaire that allows a product to be ranked and helps determine which Circular Business Model (CBM) is the best fit (Circular Business Model Opportunity Screening Tool). These tools will also be applied within ESCH-R, and at Medtronic we will certainly be using both tools as well. The collaboration was open and constructive, and her work aligns well with our ambitions. We therefore do not see this as an endpoint, but as valuable input that we will actively use in our decision-making.”
Three insights from Vita’s project!
Science in 1 – What is possible? Rethinking circularity in healthcare

Circular business models for medical devices require rethinking value, responsibility, and risk. In a highly regulated, safety-critical environment, discussions around circularity often focus on what is not allowed, limiting sustainable innovation.
Vita: “In my research, I aimed to shift this perspective by encouraging stakeholders to reflect on what is possible, allowed, or could be reconsidered. Through interviews across the medtech industry, hospitals, and waste management sectors, I examined stakeholder-specific challenges. This created an integrated view, highlighting how siloed decisions shape the wider system.”
By taking a realistic yet optimistic approach, acknowledging regulatory boundaries while actively exploring opportunities for change, this research opens up space for dialogue and collective rethinking of what circularity in healthcare can look like.
Science in 2 – Context determines what is ‘circular’ and promising.
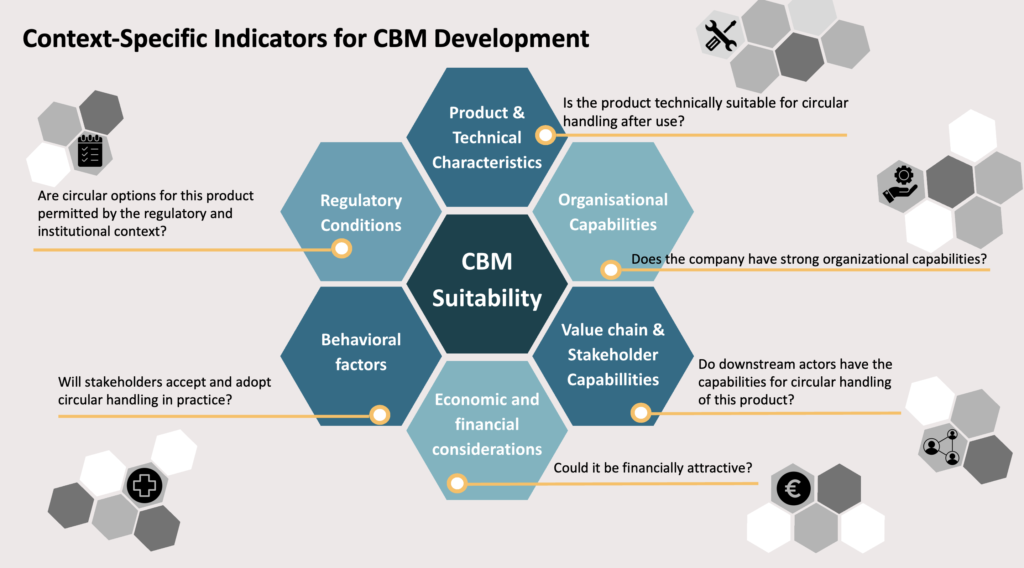
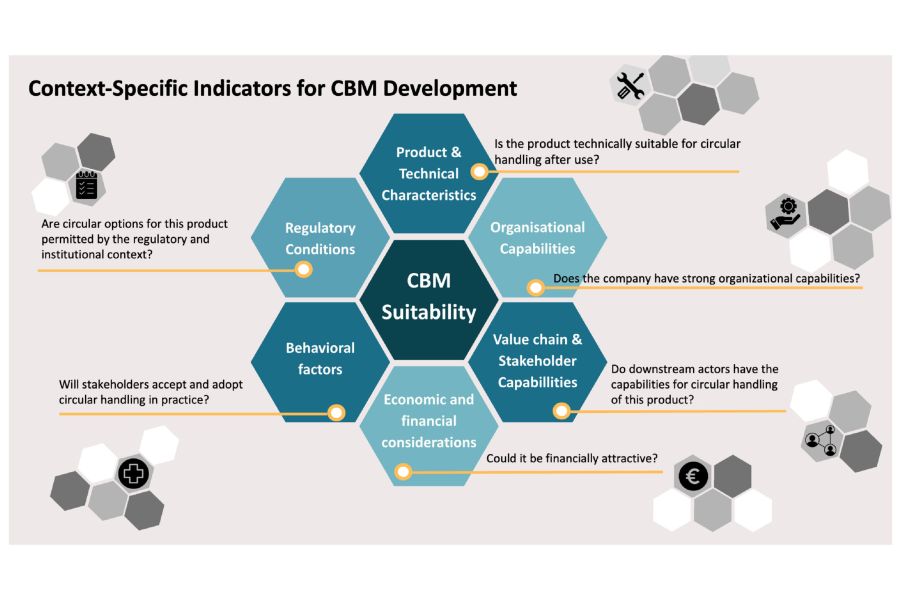
Whether a circular business model can be implemented for a medical device depends on more than the product itself; it is shaped by the broader system, including regulatory requirements, hospital infrastructure, stakeholder roles.
Vita: “At the same time, stakeholders operate within different business models, meaning that circular solutions must also be economically viable. For example, recycling often requires scale, while hospitals need infrastructure for reverse logistics. In addition, product design, such as ease of disassembly, also plays a key role in enabling circular strategies.”
Ultimately, reuse, recycling, or refurbishment depend on how multiple factors align within a specific context. Circular opportunities are therefore context-dependent, making early-stage screening essential. By bringing together diverse perspectives and combining internal and external knowledge, support organizations develop solutions suited to their environment.
Science in 3 – Collaboration as a driver of change
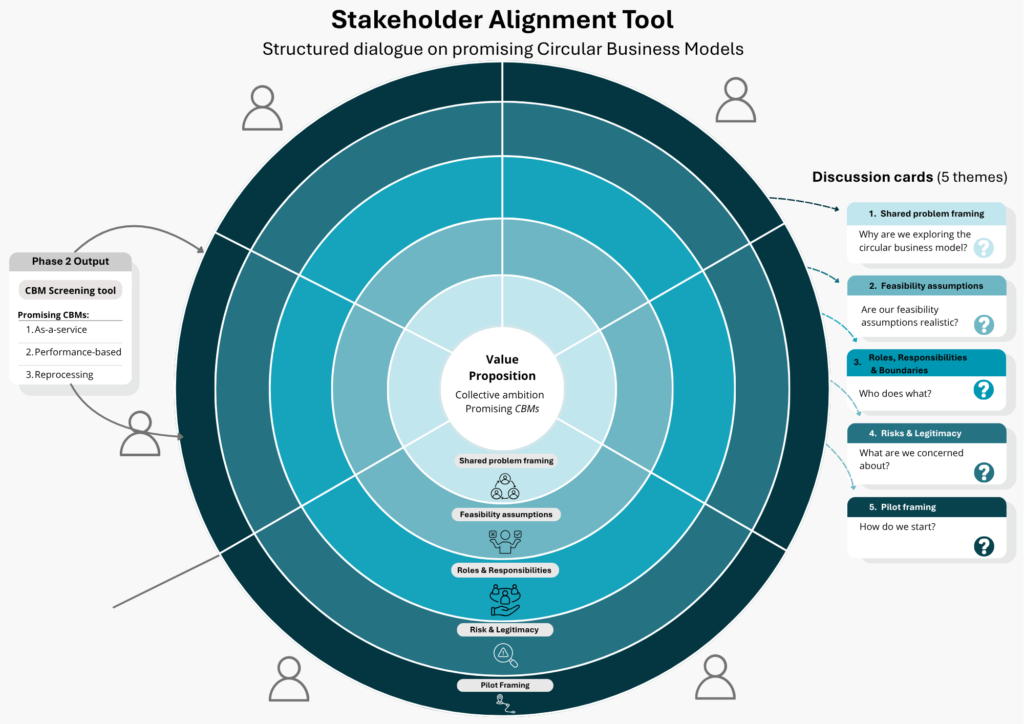
Beyond identifying opportunities, implementing circular solutions remains a collaborative effort, as the transition toward circular business models involves multiple phases and steps.
Vita: “Internal commitment serves as the starting point, followed by opportunity screening and subsequent stakeholder alignment. This process involves openly discussing assumptions, risks, roles, and responsibilities. The value lies not only in the analysis itself, but also in the dialogue it creates, making implicit tensions visible and enabling stakeholders to jointly explore potential solutions. Through stakeholder alignment, it becomes clearer which requirements are non-negotiable and where compromises can be made.”
In this way, alignment is not a passive outcome, but an active and necessary step toward implementation, with coordination or orchestration within the network helping to translate insights into concrete action.
Vita’s project was supervised by Duygu Keskin (ESCH-R WP4), Jacopo Parma (ESCH-R PhD WP4) and Janny Boekhout (Medtronic).


